Comparison of 10 Essential Benefits CA vs Federal
Medi-Cal provides benefits similar to the coverage options available through Covered California, but often at lower or no cost to you or your family. All of the health plans offered through Covered California or by Medi-Cal include the same comprehensive set of benefits known as “essential health benefits.” Essential health benefits consist of: DHCS.ca.gov
10 Essential [Mandated] Health Benefits (EBH)
under Health Care Reform, PPACA
ACA Obamacare Essential (Mandatory) Benefits
and #CA California Essential Health Benefits
- CHCF Comparison of CA and Federal Essential Health Benefits
- (A) Ambulatory patient services.
- (B) Emergency services.
- Emergency response ambulance or ambulance transport services
- (C) Hospitalization.
- (D) Maternity [but not infertility - CA Law? ] and newborn care.
- §146.130 Standards relating to benefits for mothers and newborns.
- Maternity: Inpatient hospital and ambulatory
- Prescription drug coverage for contraceptives
- Maternity hospital stay
- Sterilization operations and procedures View the Affordable health ca.com page Assembly Bill 1453 (Monning) and Senate Bill 951 (Hernandez). View the California Health and Safety Code, section 1367.005
- Abortion G-d forbid
- (E) Mental health and substance use disorder services, including behavioral health treatment.
- California Mental health parity
- Autism care (FAQ) including behavioral health treatment
- autism learning partners.com/medi-cal
- insurance.ca.gov/autism
- Sample Policy - use ctrl f to search for autism
- Medi Cal FAQ's
- Autism care (FAQ) including behavioral health treatment
- Biden administration finalizes rules to ensure insurers pay for mental health care
- California Mental health parity
- (F) Prescriptions Drugs – Rx – Including under 65 ACA CFR 156.122
- (G) Rehabilitative and habilitative [learn or improve skills for daily living] services and devices.
- (H) Laboratory services.
- (I) Preventive and wellness services and chronic disease management.
- Diabetes education, management and treatment
- (J) Pediatric services, including Dental - oral and vision care. Blue Shield Individual Flyer Essential Health Benefits 5.2013 Group Essential Health Benefits (EBH) 42 USC 18022 SB 951
- (H?) Cancer and other life threatening disease - clinical trials
- AIDS vaccine
- HIV testing
- Organ transplants for HIV
- Alpha feto protein testing
- Prosthetics for laryngectomy
- Reconstructive surgery
- Mastectomies and lymph node dissections
- Cervical cancer treatment
- Cancer screening tests
- Cancer clinical trials
- Prostate cancer treatment
- Osteoporosis treatment
- Surgical procedures for jaw bones
- Anesthesia for dental
- Conditions attributable to diethylstilbestrol
- Hospice (end of life) care
- Pain management medication for terminally ill
- Phenylketonuria treatment
- Health Care.Gov
- CMS.gov very detailed
-
White House.Gov Affordable Health Care Act YouTube Channel
- Here's the Feb. 20, 2013 final.rule establishing the essential health benefits (EHBs) for 10 categories of care, including basic services such as hospitalization and emergency care, as well as mental health and maternity care. In addition, the plans must cover a minimum of 60 percent of the actuarial value of covered medical services.
-
No more Annual & Lifetime Limits under Health Care Reform Aetna's Explanation
- § 1300.67.005. Essential Health Benefits Westlaw Barclays California Code of Regulations
- CHCF Comparison of CA and Federal Essential Health Benefits
- CA Department of Insurance
- Covered CA
- Medi Cal Listing
- California Benchmarks Kaiser Small Group HMO 30 ♦
- Updates chbrp.org * California’s Essential Health Benefits and Updating the Benchmark Plan dmhc.ca.gov *
- SB 62 Update CA Benchmark Plan Essential Benefits legislature.ca.gov 10/2025
- IMHO the easiest is to read your EOC Evidence of Coverage
- CHCF Comparison of CA vs Federal Rules as of 2014
- Frequently Asked Questions on Essential Health Benefits Bulletin from the Department of Health and Human Services (PDF)
- Essential Health Benefits Bulletin from the Department of Health and Human Services, Dec. 16, 2011 (PDF)
-
No more Annual & Lifetime Limits under Health Care Reform Aetna's Explanation
- Short Term Policies that don't have essential benefits have been banned in CA for a long time. Now the Feds are doing it. Kff.org
- Visit our webpage on Preventive services
- Our main webpage on California & Federal Essential Health Benefits
- jump to section on:
All our Health plans are Guaranteed Issue with No Pre X Clause
Instant Quote & Subsidy #Calculation
There is No charge for our complementary services, we are paid by the Insurance Company.
- Our Quote Engine Takes all the complexity out of using pencil and paper to figure out the premiums per the Obamacare/ACA rules under CFR §1.36B-3 *
- We are authorized Brokers for Dental, Vision & Covered CA get instant quotes direct and in Covered CA with subsidy calculation for:
- Watch our 10 minute VIDEO that explains everything about getting a quote
By submitting the information below , you are agreeing to be contacted by Steve Shorr a Licensed Sales Agent by email, texting or Zoom to discuss Medicare or other Insurance Plans as relevant to your inquiry. This is a solicitation for Insurance
Lab Fees
What and how much is covered for #Lab – Laboratory Fees – as an essential benefit under Health Care Reform?
- Is the $10 limit for most X-rays and laboratory test limited only to this Blue Shield Platinum plan?
- ***I believe the co-pay would depend on what Metal Level, one has purchased.
- Are the words “most” and “encounter” defined anywhere?
- ***Proactive Office Encounter is a Kaiser Term – Here’s an explanation… but I don’t find a simple definition.
- I see that essential benefits include laboratory tests for PSA, cholesterol and diabetes are no charge items as they are deemed preventative care.
- ***Yes, but see the limitation for conditions that are already diagnosed!
- Our current Blue Shield of CA PPO small business plan took effect on July 1, 2011. As I have previously explained, when I last had routine lab tests, which was in October, 2012, the cost was over $1,000 from an in-network provider. So, since the Affordable Care Act has taken more full effect, is there any chance those lab tests which are now deemed preventative care and thus no charge,
- ***I’m glad you’ve asked this question… I don’t think ANYONE knows of the limitation for what’s already been diagnosed.
- will be no charge under our current plan, or will we have to wait until a new plan is in effect?
- Office Visits
- An Office Visit is when You go to a Physician’s office and have one or more of ONLY the following three services provided:
-
History-Gathering of information on an illness or injury.
-
Examination
-
Physician’s medical decision regarding the diagnosis and treatment plan.
- Office Visit will not include any other services while at the office of a Physician (e.g., any surgery, Infusion Therapy, diagnostic X-ray, laboratory, pathology and radiology) or any services performed other than the three services specifically listed above.
- Covered Services include:
- Office Visits with Primary care Physicians and Providers (PCP) and Specialty Care Physicians and Providers
- Home Visits for medical care to examine, diagnose, and treat an illness or injury.
- Note: Physician visits in the home are different than the “Home Care Services” benefit described earlier in this Agreement (Page 88)
- benchmark.Kaiser Small Group HMO
- One might also consider these options for fees that don’t get paid
- health-savings-accounts-hsa/
- Section 105 medical_expense_reimbursement
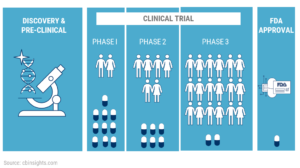
Clinical Trial
#Clinical Trials are Covered under Health Care Reform
- Health Care Reform/ACA/Obamacare requires health plans to pay the routine care costs of patients who participate in clinical trials for the prevention, detection and treatment of cancer and other life-threatening conditions. Read the entire article ⇒ Kaiser Health News – Simple Explanation 8.24.2010Aetna.com explanation of how clinical trials are an essential benefit under Health Care Reform
- Specimen Platinum Policy – Clinical Studies Page 71
- Experimental Exclusion Page 109 163
- Independent Medical Review Page 153
- Learn more About Clinical Studies including relevant History, Policies, and Laws.
- United Health Care
- CIGNA Fact Sheet
- Clinical Trials.Gov ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world.
- The problem wasn’t the costs of the clinical trial itself:
- The cancer center would pay to administer the drug and analyze the results. But if Crusoe participated in the trial, his health plan would stop covering all the other doctor visits, hospital stays, tests and treatment related to treating his cancer. Routine patient care refers to the range of medical services people with a particular diagnosis might need. It includes treatment for side effects and other medical issues that might arise as a result of the trial. Although Medicare and many private health plans already cover such costs, some plans decline to do so on the grounds that clinical trials are experimental, say experts. More than half of states require coverage of routine costs in a clinical trial, but state requirements vary. The new law sets a minimum standard. section 2709(a) *
Definitions
Life Threatening Conditions- ‘life-threatening condition’ means any disease or condition from which the likelihood of death is probable unless the course of the disease or condition is interrupted. (of an illness, situation, etc) that makes it possible that the person affected will die ‘‘§2709 (e) * Collins Dictionary.com
- Links & References
- Federal Register Ryan White Regulations Section 2695 42 USC 300ff-131
- 42 USC 300gg-8 Coverage for individuals participating in approved clinical trials
Technical
Health Insurance unfortunately is very complicated
President Trump February 27, 2017
- Thus, if we haven't simplified and explained in PLAIN ENGLISH what you are looking for: